|
suggested a neutron burst intermediate between s− and r processes which modified the products of a regular r process by the separation of stable Xe isotopes and their radioactive precursors on short time scales after supernova explosions. To explain the isotopic compositions of Xe-HL Ott and Richter et al. Xenon carried by nanodiamond separated from primitive meteorites is anomalously enriched in both heavy and light isotopes, which is reflected in the acronym Xe-HL. There were several previous observations of 131Xe and 132Xe enrichments, similar to those found in the Oklo ores, but they were either not reproducible or tentatively attributed to neutron capture reactions on Te. In a natural environment, CFF-Xe was reproducibly observed in minerals from the Oklo reactors in Gabon which still remain the only occurrence of natural nuclear reactors. Here, Xe in the ampoule was found to be enriched in 136Xe and 134Xe, complimentarily to the low temperature Xe releases from the irradiated powder itself which was depleted in these isotopes. A modified version of this experiment included the isotopic analyses of the Xe accumulated in a neutron-irradiated quartz ampoule containing U-oxide powder. 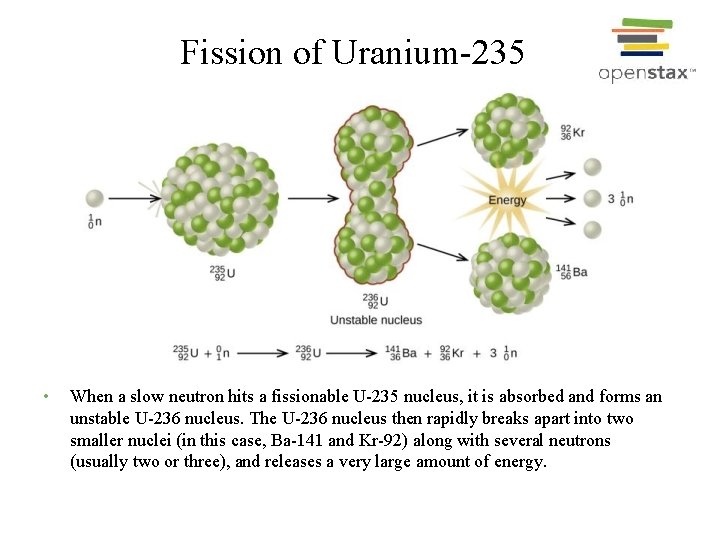
At low extraction temperatures the apparent fission yields of 131Xe and 132Xe showed ~ 10-fold enrichments. Kennett and Thode were among the first who systematically studied the formation of CFF-Xe released from irradiated U 3O 8 powder. This effect causes a separation of Xe isotopes having long and short lived β-active precursors and was called chemical fractionation fission (CFF a term proposed by Frank Podosek, confusingly close, but unrelated to, CCF, the acronym for carbonaceous chondrite fission). In the case of the pulsed neutron-induced fission, such as for the Oklo reactors, the diffusion of Xe precursors is much faster during the active fission cycle, due to the higher temperatures, than when the reactor is dormant. Therefore, the formation of different Xe isotopes is delayed from the moment when actual fission has occurred, with this delay differing for each Xe isotope. These differences allow for accurate decomposition of Xe containing both fission and nonfission isotopes.įor stable fission Xe isotopes the β − -active precursors in fission chains are the isotopes of Sn, Sb, Te, and I with half-lives ranging from seconds to 1.57 × 10 7 years for 129I. Light stable Xe isotopes Xe, Xe, and Xe are proton rich and are not produced in fission. The fission chain with M = 130 ends at nearly stable 130Te, and therefore 130Xe is effectively “shielded” from fission. Isotopes 136Xe, 134Xe, 132Xe, 131Xe, and some 129Xe are such isobars and are usually called fission Xe for short. To normalize the neutron-to-proton ratio the immediate fission fragments undergo a series of β − decays ending with a stable isobar. Xe isotopic signatures of various processes are presently well established and routinely used as genetic markers of terrestrial and extraterrestrial materials.įission fragments of heavy nuclei are neutron rich. These properties allow for accurate resolution of isotopic differences between Xe components which originated from various fissile materials, planetary atmospheres, solar wind, as well as nucleosynthetic products of s, r, and p processes in stars, while corresponding differences in other elements are barely visible. Stable xenon isotopes have high fission yields and are rare in rocks.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
